Renal cell carcinoma (RCC), as well as other solid tumours, can prove resistant to standard cancer treatments such as chemotherapy. One mechanism likely to play a role in this resistance is activation of HIF signalling either as a result of hypoxia within the tumour or as the result of mutations as in BHD, VHL and TSC. Increased HIF activity, as well as altered PI3K/AKT/mTOR, MEK/ERK and TGFβ signalling, can shift the balance of anti- and pro-apoptotic factors enabling tumour cells to survive in unfavourable conditions.
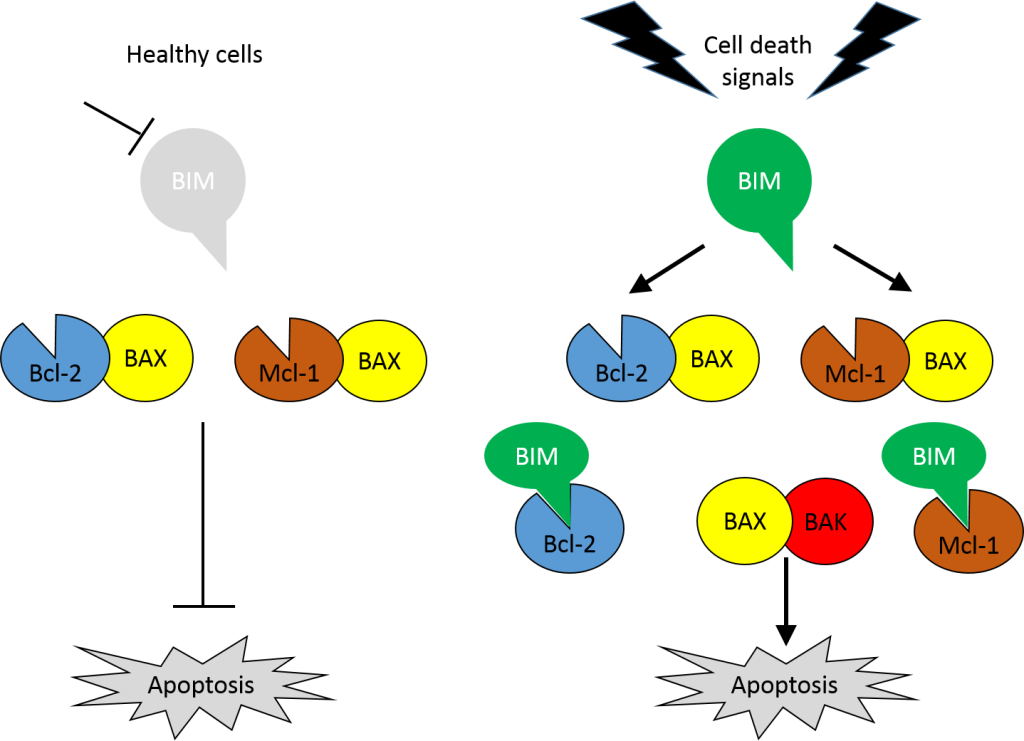
In response to cell death signals BH3-only family members, such as BIM, bind to and sequester the anti-apoptotic BCL-2 family members, liberating the pro-apoptotic BAX and BAK proteins that induce apoptosis (Figure 1). However, tumour-specific loss of BIM, and associated resistance to apoptosis, has been reported in several tumour types: in clear cell RCC samples (35/45) with a correlation between BIM expression and apoptosis-susceptibility identified in RCC lines (Zantl et al., 2007); in BHD tumour samples (Cash et al., 2011) and VHL-deficient cell lines (Guo et al., 2009) suggesting a role for FLCN and pVHL in BIM expression and stability respectively; and in breast cancer tumour lines with aberrant PI3K/AKT/mTOR or MEK/ERK signalling (Faber et al., 2011).
Figure 1: BIM binds to and sequesters Bcl-2 and Mcl-1 to induce apoptosis via dimerisation of BAX and BAK.
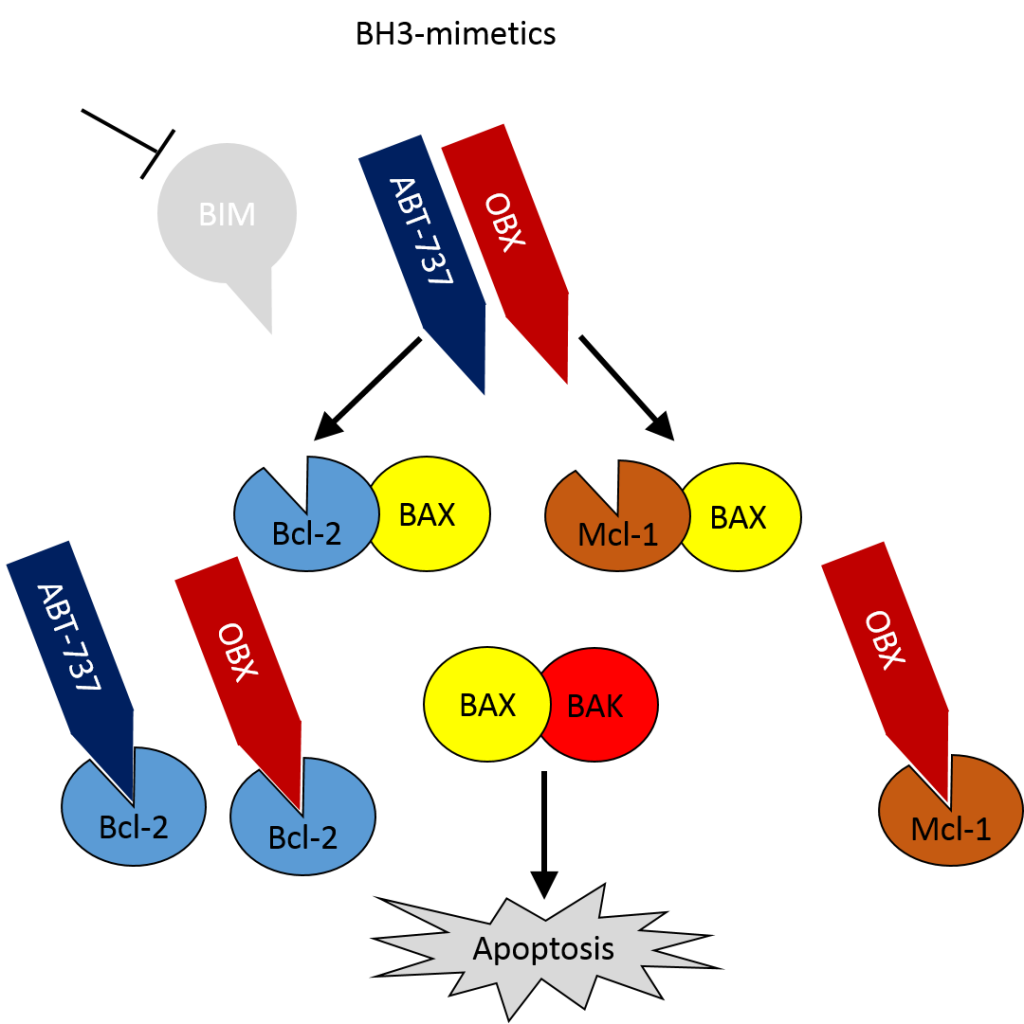
One potential treatment for such tumours is the use of BH3-mimetics – small molecule inhibitors, such as ABT-737 (or the orally bioavailable formulation ABT-263) and Obatoclax (OBX), which bind to BCL-2 and induce apoptosis (Figure 2, Oltersdorf et al., 2005, Tse et al., 2008, Nguyen et al., 2007). OBX has a broader specificity that ABT-737 binding MCL-1 as well as BCL-2, making it a more potent antagonist. Additionally it has been shown that OBX can reduce mTOR activity in melanoma cells (Espona-Fiedler et al., 2012) and both disrupt HIF-1-α protein synthesis and enhance proteosomal degradation to lower HIF-1α protein levels under hypoxic conditions (Gariboldi et al., 2015). As HIF signalling is often perturbed in RCC this suggests that the use of BH3-mimetic may also be useful in these cases.
Figure 2: BH3-mimetics can bind Bcl-2 and Mcl-1 to induce apoptosis in the absence of BIM.
BH3-mimetics could be of particular interest in BHD as several of the pathways known to be altered in FLCN-deficient cells have been implicated in the control of BIM expression: increased HIF-signalling plays a role in BHD renal and pulmonary pathology (Preston et al., 2011, Nishii et al., 2013); increased mTOR activity has been seen in patient samples and BHD models (Baba et al., 2008, Nishii et al., 2013); and increased activation of ERK1/2 signalling was identified in FLCN-null kidneys (Baba et al., 2008).
In addition to these pathways Cash et al., (2011) reported a TGFβ-dependent reduction in BIM expression and increased apoptotic resistance in Flcn-/- ES cells, with Flcn-restored cells showing rescued apoptotic response. A role for FLCN in the regulation of TGFβ signalling had previous been suggested (Hong et al., 2010) following the reduction in TGFβ-target expression in FLCN-null cells (including SMAD7 as discussed in a previous blog regarding a role of MMPs in BHD). Cash et al. determined that the deregulation of TGFβ signalling resulted in hypoacetylation of target promotors, including BIM, and reduced expression. Treatment of these cells with either ABT-737 or an HDAC inhibitor increased susceptibility to apoptosis identifying them as potential therapies in BHD.
BH3-mimetics have proven successful in a range of cancers and there are several ongoing trials assessing the efficiency of ABT-263 and OBX in the treatment of solid tumours. The reduction in BIM expression in BHD tumours and the links between FLCN-associated signalling and BIM expression suggest that BH3-mimetics could be a valid treatment in BHD. Interestingly Cash et al. reported a reduction in BIM levels in a fibrofolliculoma sample raising the possibility for BH3-mimetics to be used for multiple BHD phenotypes.
- Baba M, Furihata M, Hong SB, Tessarollo L, Haines DC, Southon E, Patel V, Igarashi P, Alvord WG, Leighty R, Yao M, Bernardo M, Ileva L, Choyke P, Warren MB, Zbar B, Linehan WM, Schmidt LS (2008). Kidney-targeted Birt-Hogg-Dube gene inactivation in a mouse model: Erk1/2 and Akt-mTOR activation, cell hyperproliferation, and polycystic kidneys. J Natl Cancer Inst. Jan 16;100(2):140-54. PubMed PMID: 18182616.
- Cash TP, Gruber JJ, Hartman TR, Henske EP, & Simon MC (2011). Loss of the Birt-Hogg-Dubé tumor suppressor results in apoptotic resistance due to aberrant TGFβ-mediated transcription. Oncogene, 30 (22), 2534-46 PMID: 21258407.
- Espona-Fiedler M, Soto-Cerrato V, Hosseini A, Lizcano JM, Guallar V, Quesada R, Gao T, Pérez-Tomás R (2012). Identification of dual mTORC1 and mTORC2 inhibitors in melanoma cells: prodigiosin vs. obatoclax. Biochem Pharmacol. Feb 15;83(4):489-96. PubMed PMID: 22155350.
- Faber AC, Corcoran RB, Ebi H, Sequist LV, Waltman BA, Chung E, Incio J, Digumarthy SR, Pollack SF, Song Y, Muzikansky A, Lifshits E, Roberge S, Coffman EJ, Benes CH, Gómez HL, Baselga J, Arteaga CL, Rivera MN, Dias-Santagata D, Jain RK, Engelman JA (2011). BIM expression in treatment-naive cancers predicts responsiveness to kinase inhibitors. Cancer Discov. Sep;1(4):352-65. PubMed PMID: 22145099.
- Gariboldi MB, Taiana E, Bonzi MC, Craparotta I, Giovannardi S, Mancini M, & Monti E (2015). The BH3-mimetic obatoclax reduces HIF-1α levels and HIF-1 transcriptional activity and sensitizes hypoxic colon adenocarcinoma cells to 5-fluorouracil. Cancer letters PMID: 25979228.
- Guo Y, Schoell MC, Freeman RS (2009). The von Hippel-Lindau protein sensitizes renal carcinoma cells to apoptotic stimuli through stabilization of BIM(EL). Oncogene. Apr 23;28(16):1864-74. PubMed PMID: 19305426.
- Hong SB, Oh H, Valera VA, Stull J, Ngo DT, Baba M, Merino MJ, Linehan WM, Schmidt LS (2010). Tumor suppressor FLCN inhibits tumorigenesis of a FLCN-null renal cancer cell line and regulates expression of key molecules in TGF-beta signaling. Mol Cancer. Jun 23;9:160. PubMed PMID: 20573232.
- Nguyen M, Marcellus RC, Roulston A, Watson M, Serfass L, Murthy Madiraju SR, Goulet D, Viallet J, Bélec L, Billot X, Acoca S, Purisima E, Wiegmans A, Cluse L, Johnstone RW, Beauparlant P, Shore GC (2007). Small molecule obatoclax (GX15-070) antagonizes MCL-1 and overcomes MCL-1-mediated resistance to apoptosis. Proc Natl Acad Sci U S A. Dec 4;104(49):19512-7. PubMed PMID: 18040043.
- Nishii T, Tanabe M, Tanaka R, Matsuzawa T, Okudela K, Nozawa A, Nakatani Y, Furuya M (2013). Unique mutation, accelerated mTOR signaling and angiogenesis in the pulmonary cysts of Birt-Hogg-Dubé syndrome. Pathol Int. Jan;63(1):45-55. PubMed PMID: 23356225.
- Oltersdorf T, Elmore SW, Shoemaker AR, Armstrong RC, Augeri DJ, Belli BA, Bruncko M, Deckwerth TL, Dinges J, Hajduk PJ, Joseph MK, Kitada S, Korsmeyer SJ, Kunzer AR, Letai A, Li C, Mitten MJ, Nettesheim DG, Ng S, Nimmer PM, O’Connor JM, Oleksijew A, Petros AM, Reed JC, Shen W, Tahir SK, Thompson CB, Tomaselli KJ, Wang B, Wendt MD, Zhang H, Fesik SW, Rosenberg SH (2005). An inhibitor of Bcl-2 family proteins induces regression of solid tumours. Nature. Jun 2;435(7042):677-81. PubMed PMID: 15902208.
- Preston RS, Philp A, Claessens T, Gijezen L, Dydensborg AB, Dunlop EA, Harper KT, Brinkhuizen T, Menko FH, Davies DM, Land SC, Pause A, Baar K, van Steensel MA, Tee AR (2011). Absence of the Birt-Hogg-Dubé gene product is associated with increased hypoxia-inducible factor transcriptional activity and a loss of metabolic flexibility. Oncogene. Mar 10;30(10):1159-73 PubMed PMID: 21057536.
- Song JH, Kandasamy K, Zemskova M, Lin YW, Kraft AS (2011). The BH3 mimetic ABT-737 induces cancer cell senescence. Cancer Res. Jan 15;71(2):506-15. PubMed PMID: 21084274.
- Tse C, Shoemaker AR, Adickes J, Anderson MG, Chen J, Jin S, Johnson EF, Marsh KC, Mitten MJ, Nimmer P, Roberts L, Tahir SK, Xiao Y, Yang X, Zhang H, Fesik S, Rosenberg SH, Elmore SW (2008). ABT-263: a potent and orally bioavailable Bcl-2 family inhibitor. Cancer Res. May 1;68(9):3421-8. PubMed PMID: 18451170.
- Zantl N, Weirich G, Zall H, Seiffert BM, Fischer SF, Kirschnek S, Hartmann C, Fritsch RM, Gillissen B, Daniel PT, Häcker G (2007). Frequent loss of expression of the pro-apoptotic protein Bim in renal cell carcinoma: evidence for contribution to apoptosis resistance. Oncogene. Oct 25;26(49):7038-48. PubMed PMID: 17486061.
- Zhu S, Cohen MB, Bjorge JD, Mier JW, Cho DC (2013). PI3K inhibition potentiates Bcl-2-dependent apoptosis in renal carcinoma cells. J Cell Mol Med. 2013 Mar;17(3):377-85. PubMed PMID: 23387989.